This section describes the basic materials used in building construction and discusses their common applications. As the world’s population increases and consumes more of the natural resources, it is incumbent upon the civil engineer to use building materials that contribute to sustaining development instead of satisfying only the short-term need. Material selection should incorporate an evaluation of the amount of energy required to produce and deliver the material to the building site. This concept of ‘‘embodied energy’’ is evolving and variable. As an example, in the Pacific Northwest lumber would have an ‘‘embodied energy’’ of 1, but in the arid Southwest transportation raises the value several points. Examples of other materials are concrete (2–3), steel (4–6), and aluminium (80). For discussion purposes, materials used in similar applications are grouped and discussed in sequence, for example, masonry materials, wood, metals, plastics, etc.
Cementitious Materials
Cementitious materials include the many products that are mixed with either water or some other liquid or both to form a cementing paste that may be formed or moulded while plastic but will set into a rigid shape. When sand is added to the paste, mortar is formed. A combination of coarse and fine aggregate (sand) added to the paste forms concrete.
Types Of Cementitious Materials
There are many varieties of cements and numerous ways of classification. One of the simplest classifications is by the chemical constituent that is responsible for the setting or hardening of the cement. On this basis, the silicate and aluminate cements, wherein the setting agents are calcium silicates and aluminates, constitute the most important group of modern cements. Included in this group are the Portland, aluminous, and natural cements.
Limes, wherein the hardening is due to the conversion of hydroxides to carbonates, were formerly widely used as the sole cementitious material, but their slow setting and hardening are not compatible with modern requirements. Hence, their principal function today is to plasticize the otherwise harsh cements and add resilience to mortars and stuccoes. Use of limes is beneficial in that their slow setting promotes healing, the recementing of hairline cracks.
Another class of cements is composed of calcined gypsum and its related products. The gypsum cements are widely used in interior plaster and for fabrication of boards and blocks; but the solubility of gypsum prevents its use in construction exposed to any but extremely dry climates.
Oxychloride cements constitute a class of specialty cements of unusual properties. Their cost prohibits their general use in competition with the cheaper cements; but for special uses, such as the production of spark proof floors, they cannot be equalled.
Masonry cements or mortar cements are widely used because of their convenience. While they are, in general, mixtures of one of more of the above-mentioned cements with some admixtures, they deserve special consideration because of their economies.
Other cementitious materials, such as polymers, fly ash, and silica fume, may be used as a cement replacement in concrete. Polymers are plastics with long-chain molecules. Concretes made with them have many qualities much superior to those of ordinary concrete.
Silica fume, also known as micro silica, is a waste product of electric-arc furnaces. The silica reacts with limes in concrete to form a cementitious material. A fume particle has a diameter only 1% of that of a cement particle.
Portland Cements
Portland cement, the most common of the modern cements, is made by carefully blending selected raw materials to produce a finished material meeting the requirements of ASTM C150 for one of eight specific cement types. Four major compounds [lime (CaO), iron (Fe2O3), silica (SiO2), and alumina (Al2O3)] and two minor compounds [gypsum (CaSO4 2H2O) and magnesia (MgO)] constitute the raw materials. The calcareous (CaO) materials typically come from limestone, calcite, marl, or shale. The argillaceous (SiO2 and Al2O3) materials are derived from clay, shale, and sand. The materials used for the manufacture of any specific cement are dependent on the manufacturing plant’s location and availability of raw materials. Portland cement can be made of a wide variety of industrial by-products.
In the manufacture of cement, the raw materials are first mined and then ground to a powder before blending in predetermined proportions. The blend is fed into the upper end of a rotary kiln heated to 2600 to 3000F by burning oil, gas, or powdered coal. Because cement production is an energy-intensive process, reheaters and the use of alternative fuel sources, such as old tires, are used to reduce the fuel cost. (Burning tires provide heat to produce the clinker and the steel belts provide the iron constituent.) Exposure to the elevated temperature chemically fuses the raw materials together into hard nodules called cement clinker. After cooling, the clinker is passed through a ball mill and ground to a fineness where essentially all of it will pass a No. 200 sieve (75 m). During the grinding, gypsum is added in small amounts to control the temperature and regulate the cement setting time. The material that exits the ball mill is Portland cement. It is normally sold in bags containing 94 lb of cement.
Concrete, the most common use for Portland cement, is a complex material consisting of Portland cement, aggregates, water, and possibly chemical and mineral admixtures. Only rarely is Portland cement used alone, such as for a cement slurry for filling well holes or for a fine grout. Therefore, it is important to examine the relationship between the various Portland cement properties and their potential effect upon the finished concrete. Portland cement concrete is generally selected for structural use because of its strength and durability. Strength is easily measured and can be used as a general directly proportional indicator of overall durability. Specific durability cannot be easily measured but can be specified by controlling the cement chemistry and aggregate properties.
Specifications for Portland Cements
ASTM C150 defines requirements for eight types of Portland cement. The pertinent chemical and physical properties are shown in Table 4.1. The chemical composition of Portland cement is expressed in a cement-chemistry shorthand based on four phase compounds: tricalcium silicate (C3S), dicalcium silicate (C2S), tricalcium aluminate (C3A), and tetra calcium aluminium ferrite (C4AF). C2S and C3S are termed the calcium silicate hydrates (CSH).
Most cements will exceed the requirements shown in Table 4.1 by a comfortable margin. Note that the required compressive strengths are minimums. Almost without exception, every Portland cement will readily exceed these minimum values. However, a caution must be attached to compressive strengths that significantly exceed the minimum values. While there is not a one-to-one correlation between a cement cube strength and the strength of concrete made with that cement (5000- psi cement does not equate to 5000-psi concrete), variations in cube strengths will be reflected in the tested concrete strength. It is imperative that, as the designed concrete strength reaches 5000 psi and greater, the cement cube strength be rigorously monitored. Any lowering of the running average will have a negative effect on the strength of concrete if the concrete mix design is not altered. The basic types of Portland cement covered by ASTM C150 are as follows:
Type I, general-purpose cement is the one commonly used for many structural purposes. Chemical requirements for this type of cement are limited to magnesia and sulphur-trioxide contents and loss on ignition, since the cement is adequately defined by its physical characteristics.
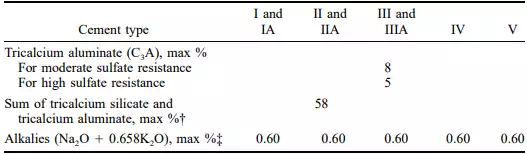
Type II is a modified cement for use in general concrete where a moderate exposure to sulphate attack may be anticipated or where a moderate heat of hydration is required. These characteristics are attained by placing limitations on the C3S and C3A content of the cement. Type II cement gains strength a little more slowly than Type I but ultimately will achieve equal strength. It is generally available in most sections of the country and is preferred by some engineers over Type I for general construction. Type II cement may also be specified as a low-alkali cement for use where alkali reactive aggregates are present. To do so requires that optional chemical requirements (Table 4.2) be included in the purchase order.
Type II low-alkali cement is commonly specified in California. Type III cement attains high early strength. In 7 days, strength of concrete made with it is practically equal to that made with Type I or Type II cement at 28 days. This high early strength is attained by finer grinding (although no minimum is placed on the fineness by specification) and by increasing the C3S and C3A content
TABLE 4.1 Chemical and Physical Requirements for Portland Cement*
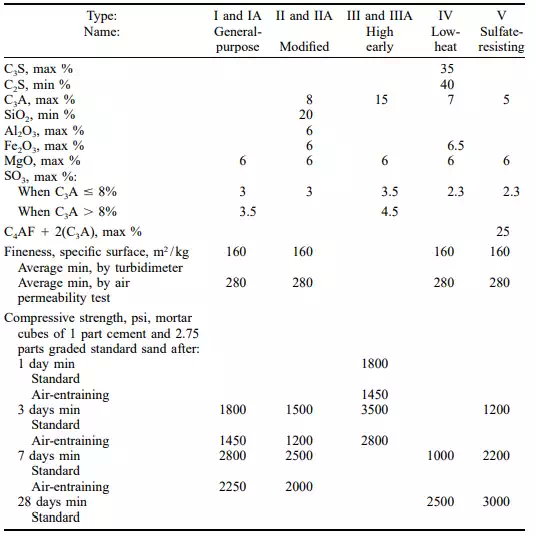
of the cement. Type III cement, however, has high heat evolution and therefore should not be used in large masses. Because of the higher C3A content, Type III cement also has poor sulphate resistance. Type III cement is not always available from building materials dealers’ stocks but may be obtained by them from the cement manufacturer on short notice. Ready-mix concrete suppliers generally do not stock Type III cement because its shorter set time makes it more volatile to transport and discharge, especially in hot weather.
Type IV is a low-heat cement that has been developed for mass concrete construction. Normal Type I cement, if used in large masses that cannot lose heat by radiation, will liberate enough heat during the hydration of the cement to raise the temperature of the concrete as much as 50 or 60F. This results in a relatively large increase in dimensions while the concrete is still soft and plastic. Later, as the concrete cools are hardening, shrinkage causes cracks to develop, weakening the concrete and affording points of attack for aggressive solutions. The potential-phase compounds that make the largest contribution to the heat of hydration are C3S and C3A; so the amounts of these are permitted to be present are limited. Since these compounds also produce the early strength of cement, the limitation results in a cement that gains strength relatively slowly. This is of little importance, however, in the mass concrete for which this type of cement is designed.
TABLE 4.2 Optional Chemical Requirements for Portland Cement*
Type V is a Portland cement intended for use when high sulphate resistance is required. Its resistance to sulphate attack is attained through the limitation on the C3A content. It is particularly suitable for structures subject to attack by liquors containing sulphates, such as liquids in wastewater treatment plants, seawaters, and some other natural waters.
Both Type IV and Type V cements are specialty cements. They are not normally available from dealer’s stock but are usually obtainable for use on a large job if arrangements are made with the cement manufacturer in advance.
Air-Entraining Portland Cements
For use in the manufacturer of air-entraining concrete, agents may be added to the cement by the manufacturer, thereby producing air-entraining Portland cements (‘‘Air-Entraining Additions for Use in the Manufacture of Air-Entraining Portland Cement,’’ ASTM C226). These cements are available as Types IA, IIA, and IIIA.
Aluminous Cements
These are prepared by fusing a mixture of aluminous and calcareous materials (usually bauxite and limestone) and grinding the resultant product to a fine powder. These cements are characterized by their rapid-hardening properties and the high strength developed at early ages. Table 4.3 shows the relative strengths of 4-in cubes of 1:2:4 concrete made with normal Portland, high-early-strength Portland, and aluminous cements.
Since a large amount of heat is liberated with rapidly by aluminous cement during hydration, care must be taken not to use the cement in places where this
TABLE 4.3 Relative Strengths of Concrete Made from Portland and Aluminous Cements*
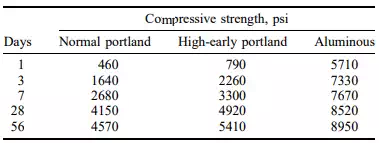
heat cannot be dissipated. It is usually not desirable to place aluminous-cement concretes in lifts of over 12 in; otherwise the temperature rise may cause serious weakening of the concrete.
Aluminous cements are much more resistant to the action of sulphate waters than are Portland cements. They also appear to be much more resistant to attack by water containing aggressive carbon dioxide or weak mineral acids than the silicate cements. Their principal use is in concretes where advantage may be taken of their very high early strength or of their sulphate resistance, and where the extra cost of the cement is not an important factor.
Another use of aluminous cements is in combination with firebrick to make refractory concrete. As temperatures are increased, dehydration of the hydration products occurs. Ultimately, these compounds create a ceramic bond with the aggregates.
Natural Cements
Natural cements are formed by calcining a naturally occurring mixture of calcareous and argillaceous substances at a temperature below that at which sintering takes place. The ‘‘Specification for Natural Cement,’’ ASTM C10, requires that the temperature be no higher than necessary to drive off the carbonic acid gas. Since natural cements are derived from naturally occurring materials and no particular effort is made to adjust the composition, both the composition and properties vary rather widely. Some natural cements may be almost the equivalent of Portland cement in properties; others are much weaker. Natural cements are principally used in masonry mortars and as an admixture in Portland-cement concretes.

